Flow Cytometry in the Diagnostic Laboratory Workup of Acute Lymphoblastic Leukemias
CC BY 4.0 · Indian J Med Paediatr Oncol 2023; 44(05): 474-481
DOI: DOI: 10.1055/s-0043-1772204
Abstract
Acute lymphoblastic leukemias (ALLs) are hematological neoplasms characterized by clonal proliferation of lymphoid blasts, which can be B- or T-cell type. Flow cytometric immunophenotyping is an integral component in establishing blast lineage during the diagnostic workup of ALLs, aiding in appropriate therapy, prognostication, and monitoring of the disease. The current review focuses on the utility of flow cytometry in the workup of ALLs, including the usefulness of various antibodies and pitfalls in diagnosis.
Keywords
acute lymphoblastic leukemia - flow cytometry - B-ALL - T-ALL
Publication History
Article published online:
04 November 2023
© 2023. The Author(s). This is an open access article published by Thieme under the terms of the Creative Commons Attribution License, permitting unrestricted use, distribution, and reproduction so long as the original work is properly cited. (https://creativecommons.org/licenses/by/4.0/)
Thieme Medical and Scientific Publishers Pvt. Ltd.
A-12, 2nd Floor, Sector 2, Noida-201301 UP, India
Abstract
Acute lymphoblastic leukemias (ALLs) are hematological neoplasms characterized by clonal proliferation of lymphoid blasts, which can be B- or T-cell type. Flow cytometric immunophenotyping is an integral component in establishing blast lineage during the diagnostic workup of ALLs, aiding in appropriate therapy, prognostication, and monitoring of the disease. The current review focuses on the utility of flow cytometry in the workup of ALLs, including the usefulness of various antibodies and pitfalls in diagnosis.
Keywords
acute lymphoblastic leukemia - flow cytometry - B-ALL - T-ALL
Introduction
Multiparametric flow cytometry is an indispensable tool for the diagnosis and subclassification of acute lymphoblastic leukemia (ALL). Accurate classification of ALLs into B- or T-cell types is crucial for the optimal choice of therapeutic regimens that varies based on the ALL subtype. The antigenic expression profile, particularly the immunophenotypic aberrancies by the blasts deviating from those encountered during normal hematopoiesis, aids in the differentiation of the blasts from their normal benign counterparts. The panel of antigens for clinical testing has evolved from 4 to 13 colors or more thanks to the substantial development in antibody clones, the fluorochrome conjugate options, and a wide variety of lasers that have dramatically increased the number of antigens that can be simultaneously studied. In this article, we attempt to discuss the strategy and approach to the classification of ALL into B- or T-cell subtypes and the evolution of consensus groups for antigen/antibody/fluorochrome selection, choice of reagents, sample processing methodology for appropriate diagnosis, and classification.
B-Acute Lymphoblastic Leukemias
B-lymphoblastic leukemia/lymphoma or B-cell precursor acute lymphoblastic leukemia (BCP-ALL) is the most common malignancy seen in childhood. Approximately 75-% of BCP-ALL cases occur in children under 6 years of age.[1] However, it shows a bimodal age distribution, with a small peak occurring during the fifth decade of life.[2] BCP-ALL is diagnosed by morphology combined with immunophenotyping, typically done by multiparametric flow cytometer (MFC). Immunophenotyping is essential for differentiating BCP-ALL from acute leukemias of other lineages, like T-lymphoblastic leukemia (T-ALL), acute myeloid leukemia (AML), mixed phenotype acute leukemia (MPAL), etc.
The flow cytometric diagnosis of BCP-ALL is based on identifying an expanded population of immature B cells showing immunophenotypic aberrancies that help distinguish the leukemic blasts from normal B-cell precursors, or hematogones.[3] These aberrancies are in the form of increased or decreased intensity of expression of an antigen on the leukemic blasts compared to the normal counterparts or gain of antigen expression, which are not specific to B-lineage.[4] Knowledge of the spectrum of antigenic expression on the B-progenitor cells during development is essential to differentiate them from B-leukemic blasts.[5] The normal B-progenitors show the expression of certain antigens in a sequential, tightly regulated manner.[3] The B-progenitor cells are derived from common lymphoid progenitor cells in the bone marrow, and they undergo three stages of maturation to become mature B-lymphoid cells. These three stages of cells are called hematogones type I (early/pre-B-I), hematogones type II (intermediate/pre-B-II), and hematogones type III (late/transitional B-cells).[6] The three stages of hematogones show a stepwise increase in the intensity of expression of CD45 and CD20 while showing a stepwise decrease in intensity of CD34 and CD10, which are finally lost at the late stage. CD19 expression is the lowest in stage I, and increases significantly in stage II, with an eventual mild reduction in mature B cells. Mature B cells show loss of CD34, CD10, and TdT, bright expression of CD20 and CD45, and a surface expression of polytypic immunoglobulins.[5] [Fig. 1] shows flow cytometric dot plots of a BCP-ALL patient.
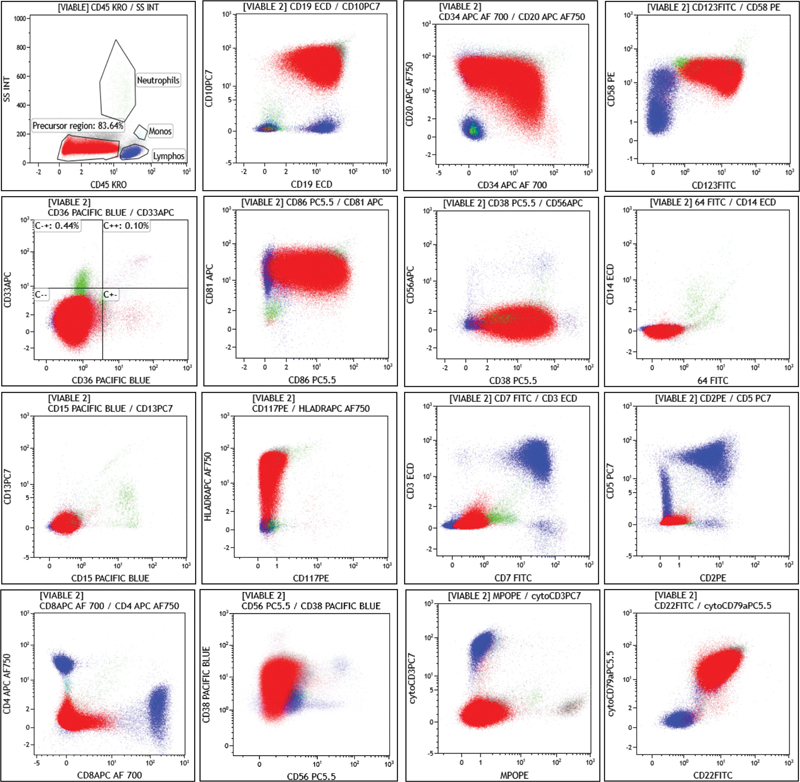
| Figure 1:Flow cytometric dot plots of a case of BCP-ALL. The blasts (red population) are SSC low, CD45 dim to negative, positive for CD19, CD10, CD34, CD20, CD58, CD123, CD81, CD86, CD38, HLA-DR, CD22, and CytoCD79a. T-cell markers like CD3, CD7, CD5, CD2, CD4, CD8 and NK cell marker like CD56, and, myeloid markers like CD13, CD15, CD33, CD26, CD117, CD14, CD64 and MPO are negative.
The B-leukemic blasts show maturation arrest at any stage and deviation from the normal sequential immunophenotypic maturation kinetics seen in hematogones.[6] [7] [8] Other than this, over- or underexpression of markers like CD123, CD58, CD81, CD86, CD73, CD200, CD9, and CD304 can be seen in the B-leukemic blasts, as shown in [Table 1]. Additionally, they may express aberrant lineage markers like CD13, CD33, CD66c, CD15, CD56, and CD7 that aid in differentiating the B-leukemic blasts from hematogones.[4] [6] [7] [9] [10] [11] [12] [13] [14] [15] [16] [17]
|
Antigen |
Aberrancy |
|---|---|
|
TdT |
Negative/uniform expression |
|
CD34 |
Negative/uniform expression |
|
CD45 |
Negative/uniform expression |
|
CD10 |
Negative/over[removed]uniform bright) |
|
CD20 |
Uniform expression or negative |
|
CD22 |
Negative/under expression |
|
CD38 |
Under expression |
|
CD19 |
Under/overexpression |
|
CD73 |
Overexpression |
|
CD58 |
Overexpression |
|
CD86 |
Overexpression |
|
CD123 |
Overexpression |
|
CD200 |
Overexpression |
|
CD81 |
Under expression |
|
CD304 |
Aberrant expression |
|
CD9 |
Overexpression |
|
CD44 |
Overexpression |
|
CD13, CD33, CD66c, CD15, CD7, and CD56 |
Lineage aberrant markers |
|
Subclassification |
CD10 |
Cytoplasmic IgM |
Surface IgM |
|---|---|---|---|
|
B I (pro-B) ALL |
Negative |
Negative |
Negative |
|
B II (pre-pre-B or common B) ALL |
Positive |
Negative |
Negative |
|
B III (pre-B) ALL |
Positive |
Positive |
Negative |
|
B IV (mature B) ALL |
Negative/positive |
Negative/positive |
Positive |
|
Tubes |
Fluorochromes |
|||||||
|---|---|---|---|---|---|---|---|---|
|
PacB |
AmCyan |
FITC |
PE |
PerCPCy5.5 |
PECy7 |
APC |
AF700 |
|
|
ALOT Tube |
CyCD3 |
CD45 |
CyMPO |
CyCD79a |
CD34 |
CD19 |
CD7 |
SmCD3 |
|
BCP-ALL Tube 1 |
CD20 |
CD45 |
CD58 |
CD66c |
CD34 |
CD19 |
CD10 |
CD38 |
|
BCP-ALL Tube 2 |
SmIgk |
CD45 |
CyIgμ |
CD33 |
CD34 |
CD19 |
SmIgM and CD117 |
SmIgλ |
|
BCP-ALL Tube 3 |
CD9 |
CD45 |
NuTdT |
CD13 |
CD34 |
CD19 |
CD22 |
CD24 |
|
BCP-ALL Tube 4 |
CD21 |
CD45 |
CD15 and CD65 |
NG2 |
CD34 |
CD19 |
CD12 3 |
CD81 |
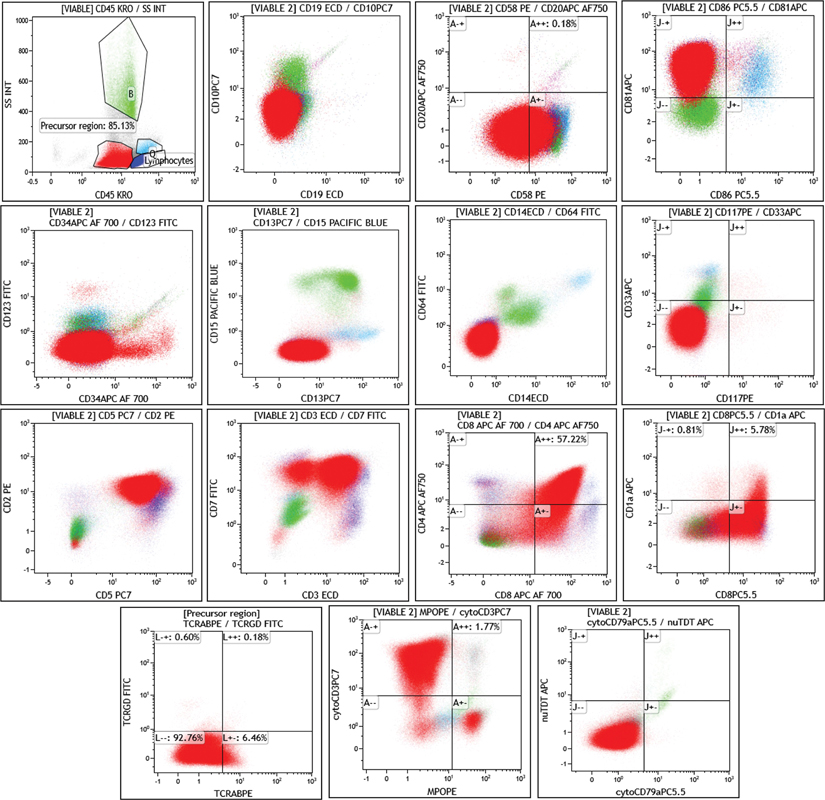
| Figure 2:Flow cytometric dot plots of a case of T-ALL. The blasts (red population) are SSC low and CD45 dim, positive forcytoCD3, CD2, CD5, smCD3, CD7, CD4, CD8, and negative for CD1a, TCRab, TCRgd, and nuTdT. The blasts are negative for B-cell and myeloid markers.
|
Markers |
Pro-T-ALL |
Pre-T-ALL |
Cortical T-ALL |
Medullary T-ALL |
|---|---|---|---|---|
|
CD1 |
– |
– |
+ + |
– |
|
CD2 |
+ |
+ + |
+ + |
+ + |
|
SmCD3 |
– |
– |
– (except in SmCD3+ subtypes, ++) |
+ + |
|
CyCD3 |
+ + |
+ + |
+ + |
+ + |
|
CD4–/CD8– |
+ + |
+ |
– |
– |
|
CD4–/CD8+ |
– |
± |
± |
± |
|
CD4+/CD8– |
– |
± |
± |
+ |
|
CD4+/CD8+ |
– |
– |
+ |
± |
|
CD5 |
– |
+ + |
+ + |
+ + |
|
CD7 |
+ + |
+ + |
+ + |
+ + |
|
T-ALL tubes |
Fluorochromes |
|||||||
|---|---|---|---|---|---|---|---|---|
|
PacB |
AmCyan |
FITC |
PE |
PerCPCy5.5 |
PECy7 |
APC |
AF700 |
|
|
Tube 1 |
CyCD3 |
CD45 |
NuTdT |
CD99 |
CD5 |
CD10 |
CD1a |
SmCD3 |
|
Tube 2 |
CyCD3 |
CD45 |
CD2 |
CD117 |
CD4 |
CD8 |
CD7 |
SmCD3 |
|
Tube 3 |
CyCD3 |
CD45 |
TCRgd |
TCRab |
CD33 |
CD56 |
CyTCRb |
SmCD3 |
|
Tube 4 |
CyCD3 |
CD45 |
CD44 |
CD13 |
HLADR |
CD45RA |
CD123 |
SmCD3 |

| Figure 1:Flow cytometric dot plots of a case of BCP-ALL. The blasts (red population) are SSC low, CD45 dim to negative, positive for CD19, CD10, CD34, CD20, CD58, CD123, CD81, CD86, CD38, HLA-DR, CD22, and CytoCD79a. T-cell markers like CD3, CD7, CD5, CD2, CD4, CD8 and NK cell marker like CD56, and, myeloid markers like CD13, CD15, CD33, CD26, CD117, CD14, CD64 and MPO are negative.

| Figure 2:Flow cytometric dot plots of a case of T-ALL. The blasts (red population) are SSC low and CD45 dim, positive forcytoCD3, CD2, CD5, smCD3, CD7, CD4, CD8, and negative for CD1a, TCRab, TCRgd, and nuTdT. The blasts are negative for B-cell and myeloid markers.
Abbreviation: T-ALL, T-acute lymphoblastic leukemia.
References
- 1 Gujral S, Badrinath Y, Kumar A. et al. Immunophenotypic profile of acute leukemia: critical analysis and insights gained at a tertiary care center in India. Cytometry B Clin Cytom 2009; 76 (03) 199-205
- 2 Paul S, Kantarjian H, Jabbour EJ. Adult acute lymphoblastic leukemia. Mayo Clin Proc 2016; 91 (11) 1645-1666
- 3 Wood BL. Flow cytometry in the diagnosis and monitoring of acute leukemia in children. J Hematop 2015; 8: 191-199
- 4 Rezaei MS, Esfandiari N, Refoua S, Shamaei M. Characterization of immunophenotypic aberrancies in adult and childhood acute lymphoblastic leukemia: lessons from regional variation. Iran J Pathol 2020; 15 (01) 1-7
- 5 DiGiuseppe JA. . Acute lymphoblastic leukemia/lymphoma: diagnosis and minimal residual disease detection by flow cytometric immunophenotyping. In: Detrick B, Schmitz JL, Hamilton RG, eds. Manual of Molecular and Clinical Laboratory Immunology. Washington, DC: ASM Press; 2016
- 6 Sędek Ł, Bulsa J, Sonsala A. et al. The immunophenotypes of blast cells in B-cell precursor acute lymphoblastic leukemia: how different are they from their normal counterparts?. Cytometry B Clin Cytom 2014; 86 (04) 329-339
- 7 McKenna RW, Washington LT, Aquino DB, Picker LJ, Kroft SH. Immunophenotypic analysis of hematogones (B-lymphocyte precursors) in 662 consecutive bone marrow specimens by 4-color flow cytometry. Blood 2001; 98 (08) 2498-2507
- 8 van Dongen JJM, Lhermitte L, Böttcher S. et al; EuroFlow Consortium (EU-FP6, LSHB-CT-2006-018708). EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia 2012; 26 (09) 1908-1975
- 9 Thalhammer-Scherrer R, Mitterbauer G, Simonitsch I. et al. The immunophenotype of 325 adult acute leukemias: relationship to morphologic and molecular classification and proposal for a minimal screening program highly predictive for lineage discrimination. Am J Clin Pathol 2002; 117 (03) 380-389
- 10 Seegmiller AC, Kroft SH, Karandikar NJ, McKenna RW. Characterization of immunophenotypic aberrancies in 200 cases of B acute lymphoblastic leukemia. Am J Clin Pathol 2009; 132 (06) 940-949
- 11 Gupta M, Monga L, Mehrotra D, Chhabra S, Singhal S, Sen R. Immunophenotypic aberrancies in acute leukemia: a tertiary care centre experience. Oman Med J 2021; 36 (01) e218-e218
- 12 Kulis J, Sędek Ł, Słota Ł, Perkowski B, Szczepański T. Commonly assessed markers in childhood BCP-ALL diagnostic panels and their association with genetic aberrations and outcome prediction. Genes (Basel) 2022; 13 (08) 1374
- 13 Chernysheva O, Grivtsova LY, Popa A, Tupitsyn NN. . B-cell precursors: immunophenotypic features in the detection of minimal residual disease in acute leukemia. In: Aribi M, ed. Normal and Malignant B-Cell [Internet]. IntechOpen; 2020. Accessed June 20, 2022 at: https://www.intechopen.com/books/normal-and-malignant-b-cell/b-cell-precursors-immunophenotypic-features-in-the-detection-of-minimal-residual-disease-in-acute-le
- 14 Solly F, Angelot F, Garand R. et al. CD304 is preferentially expressed on a subset of B-lineage acute lymphoblastic leukemia and represents a novel marker for minimal residual disease detection by flow cytometry. Cytometry A 2012; 81 (01) 17-24
- 15 Sherif L, Azab M, Al-Akad G, Zakaria M, Atfy M, Sorour S. Cluster of differentiation 97 as a biomarker for the detection of minimal residual disease in common acute lymphoblastic leukemia. Egypt J Haematol. 2017; 42: 81-87
- 16 Kamazani FM, Bahoush GR, Aghaeipour M, Vaeli S, Amirghofran Z. CD44 and CD27 expression pattern in B cell precursor acute lymphoblastic leukemia and its clinical significance. Med Oncol 2013; 30 (01) 359
- 17 DiGiuseppe JA, Fuller SG, Borowitz MJ. Overexpression of CD49f in precursor B-cell acute lymphoblastic leukemia: potential usefulness in minimal residual disease detection. Cytometry B Clin Cytom 2009; 76 (02) 150-155
- 18 Bene MC, Castoldi G, Knapp W. et al; European Group for the Immunological Characterization of Leukemias (EGIL). Proposals for the immunological classification of acute leukemias. Leukemia 1995; 9 (10) 1783-1786
- 19 Guillaume N, Penther D, Vaida I. et al. CD66c expression in B-cell acute lymphoblastic leukemia: strength and weakness. Int J Lab Hematol 2011; 33 (01) 92-96
- 20 Owaidah TM, Rawas FI, Al Khayatt MF, Elkum NB. Expression of CD66c and CD25 in acute lymphoblastic leukemia as a predictor of the presence of BCR/ABL rearrangement. Hematol Oncol Stem Cell Ther 2008; 1 (01) 34-37
- 21 Suggs JL, Cruse JM, Lewis RE. Aberrant myeloid marker expression in precursor B-cell and T-cell leukemias. Exp Mol Pathol 2007; 83 (03) 471-473
- 22 Kulis J, Wawrowski Ł, Sędek Ł. et al. Machine learning based analysis of relations between antigen expression and genetic aberrations in childhood B-cell precursor acute lymphoblastic leukaemia. J Clin Med 2022; 11 (09) 2281
- 23 Hrusák O, Porwit-MacDonald A. Antigen expression patterns reflecting genotype of acute leukemias. Leukemia 2002; 16 (07) 1233-1258
- 24 Schwartz S, Rieder H, Schläger B, Burmeister T, Fischer L, Thiel E. Expression of the human homologue of rat NG2 in adult acute lymphoblastic leukemia: close association with MLL rearrangement and a CD10(-)/CD24(-)/CD65s(+)/CD15(+) B-cell phenotype. Leukemia 2003; 17 (08) 1589-1595
- 25 Tsagarakis NJ, Papadhimitriou SI, Pavlidis D. et al. Flow cytometric predictive scoring systems for common fusions ETV6/RUNX1, BCR/ABL1, TCF3/PBX1 and rearrangements of the KMT2A gene, proposed for the initial cytogenetic approach in cases of B-acute lymphoblastic leukemia. Int J Lab Hematol 2019; 41 (03) 364-372
- 26 Djokic M, Björklund E, Blennow E, Mazur J, Söderhäll S, Porwit A. Overexpression of CD123 correlates with the hyperdiploid genotype in acute lymphoblastic leukemia. Haematologica 2009; 94 (07) 1016-1019
- 27 Vetter T, Borowski A, Wohlmann A. et al. Blockade of thymic stromal lymphopoietin (TSLP) receptor inhibits TSLP-driven proliferation and signalling in lymphoblasts from a subset of B-precursor ALL patients. Leuk Res 2016; 40: 38-43
- 28 Pastorczak A, Sedek L, Braun M. et al. Surface expression of cytokine receptor-like factor 2 increases risk of relapse in pediatric acute lymphoblastic leukemia patients harboring IKZF1 deletions. Oncotarget 2018; 9 (40) 25971-25982
- 29 Tembhare P, Badrinath Y, Ghogale S. et al. A novel and easy FxCycle™ violet based flow cytometric method for simultaneous assessment of DNA ploidy and six-color immunophenotyping. Cytometry A 2016; 89 (03) 281-291
- 30 Gupta N, Parihar M, Banerjee S. et al. FxCycle™ based ploidy correlates with cytogenetic ploidy in B-cell acute lymphoblastic leukemia and is able to detect the aneuploid minimal residual disease clone. Cytometry B Clin Cytom 2019; 96 (05) 359-367
- 31 Thomas DA, O'Brien S, Faderl S. et al. Chemoimmunotherapy with a modified hyper-CVAD and rituximab regimen improves outcome in de novo Philadelphia chromosome-negative precursor B-lineage acute lymphoblastic leukemia. J Clin Oncol 2010; 28 (24) 3880-3889
- 32 Maury S, Chevret S, Thomas X. et al; for GRAALL. Rituximab in B-lineage adult acute lymphoblastic leukemia. N Engl J Med 2016; 375 (11) 1044-1053
- 33 Jabbour E, Richard-Carpentier G, Sasaki Y. et al. Hyper-CVAD regimen in combination with ofatumumab as frontline therapy for adults with Philadelphia chromosome-negative B-cell acute lymphoblastic leukaemia: a single-arm, phase 2 trial. Lancet Haematol 2020; 7 (07) e523-e533
- 34 Jasinski S, De Los Reyes FA, Yametti GC, Pierro J, Raetz E, Carroll WL. Immunotherapy in pediatric B-cell acute lymphoblastic leukemia: advances and ongoing challenges. Paediatr Drugs 2020; 22 (05) 485-499
- 35 Guru Murthy GS, Pondaiah SK, Abedin S, Atallah E. Incidence and survival of T-cell acute lymphoblastic leukemia in the United States. Leuk Lymphoma 2019; 60 (05) 1171-1178
- 36 Craig FE, Foon KA. Flow cytometric immunophenotyping for hematologic neoplasms. Blood 2008; 111 (08) 3941-3967
- 37 DiGiuseppe JA, Wood BL. Applications of flow cytometric immunophenotyping in the diagnosis and posttreatment monitoring of B and T lymphoblastic leukemia/lymphoma. Cytometry B Clin Cytom 2019; 96 (04) 256-265
- 38 van Dongen JJ, Krissansen GW, Wolvers-Tettero IL. et al. Cytoplasmic expression of the CD3 antigen as a diagnostic marker for immature T-cell malignancies. Blood 1988; 71 (03) 603-612
- 39 Gujral S, Tembhare P, Badrinath Y, Subramanian PG, Kumar A, Sehgal K. Intracytoplasmic antigen study by flow cytometry in hematolymphoid neoplasm. Indian J Pathol Microbiol 2009; 52 (02) 135-144
- 40 Hashimoto M, Yamashita Y, Mori N. Immunohistochemical detection of CD79a expression in precursor T cell lymphoblastic lymphoma/leukaemias. J Pathol 2002; 197 (03) 341-347
- 41 Coustan-Smith E, Mullighan CG, Onciu M. et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol 2009; 10 (02) 147-156
- 42 Sin CF, Man PM. Early T-cell precursor acute lymphoblastic leukemia: diagnosis, updates in molecular pathogenesis, management, and novel therapies. Front Oncol 2021; 11: 750789
- 43 Falcão RP, Garcia AB. Expression of CD45RA (naive) and CD45RO (memory) antigens in T-acute lymphoblastic leukaemia. Br J Haematol 1993; 85 (03) 483-486
- 44 Cavalcanti Júnior GB, Savino W, Pombo-de-Oliveira MS. CD44 expression in T-cell lymphoblastic leukemia. Braz J Med Biol Res 1994; 27 (09) 2259-2266
- 45 Lhermitte L, de Labarthe A, Dupret C. et al. Most immature T-ALLs express Ra-IL3 (CD123): possible target for DT-IL3 therapy. Leukemia 2006; 20 (10) 1908-1910
- 46 Khoury JD, Solary E, Abla O. et al. The 5th edition of the World Health Organization classification of haematolymphoid tumours: myeloid and histiocytic/dendritic neoplasms. Leukemia 2022; 36: 1703-1719
- 47 Bayón-Calderón F, Toribio ML, González-García S. Facts and challenges in immunotherapy for T-cell acute lymphoblastic leukemia. Int J Mol Sci 2020; 21 (20) 7685
- 48 Caracciolo D, Mancuso A, Polerà N. et al. The emerging scenario of immunotherapy for T-cell acute lymphoblastic leukemia: advances, challenges and future perspectives. Exp Hematol Oncol 2023; 12 (01) 5


 PDF
PDF  Views
Views  Share
Share

